A mysterious cell process named anastasis (Greek for "rising to life") challenges our idea of life being a linear march towards death, and suggests that cell death can actually be reversed under certain conditions—essentially allowing cells to un-die.Even as the cell is shrivelling up in response to radiation, toxins, or other stresses, it can in some cases undo the dying process and repair itself if the stress is taken away before the cell is completely gone, said cell biologist Denise Montell of the University of California, Santa Barbara.
Advertisement
"In the field of people studying apoptosis—this programmed cell suicide mechanism—it has been a tenet in that field that once cells trigger this death process, it's irreversible," Montell told me over the phone. Her research, beginning with a paper published by the journal Molecular Biology of the Cell in 2012, shows otherwise.Montell's lab wants to see if they can use anastasis to salvage hard-to-replace cells in the human body, which could be important in treating ischemia or heart attacks. But it could also provide an accidental, chilling glimpse into the hows and whys of cancer.
Every day, the billions of cells in our bodies actively decide whether they should continue to live, or die. Damaged cells must die—otherwise we might get cancer or other diseases—through programmed cell death processes, the most famous of which is apoptosis (from the Greek for "falling off")."There are many cells that we don't want to die. This is particularly true for the neurons in our brain, which have to last our whole life, or the cells for our heart," Montell explained. A careful balance must be struck: if too many cells die we'd develop diseases like Alzheimer's or Parkinson's, a hallmark of which is neuronal cell death.Once apoptosis begins, a critical molecule called executioner caspase is activated within the cell. It does exactly what it sounds like. The caspase goes around the cell, dicing up cell parts, and the cell starts to shrivel up. "Eventually it will break into little pieces, and then other cells come and gobble up the little pieces," she said.
Advertisement
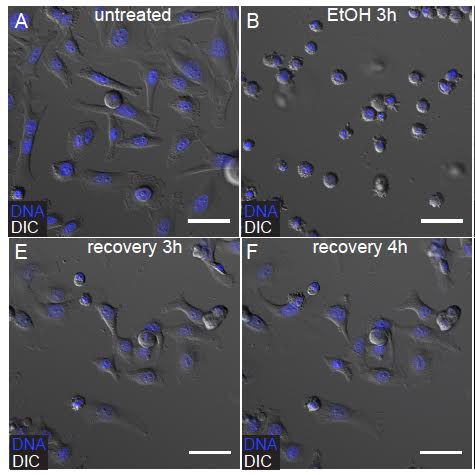
Montell began her research on anastasis years ago at John Hopkins University, where a student applied to join her lab as a postdoc after discovering the death-reversal process during his graduate work in Hong Kong. Montell credits this student—Hogan Tang—with discovering anastasis, which was aptly named with the help of classics scholars, she said.Read More: Zombie Cancer Cells Are A ProblemTang induced apoptosis in cells by adding 4.3 per cent ethanol to cell cultures, then carefully removing the toxin, Montell said. "A substantial fraction of [the cells] actually recovered."The next step was to test it on mammalian and fruit fly cells. Montell told Tang as much when he came over to visit her in 2008, and brought up what he'd observed. She was skeptical. But after several months of questioning, Tang joined her lab and they began to seriously pursue the discovery.So far, they've seen anastasis in 12 different types of mammalian cells. The fact that it can be observed in human and fruit fly cells, Montell said, suggests that anastasis is an incredibly ancient process shared by a common ancestor that must date back millions of years.There are still many unanswered questions about the ramifications and origin of the process. Andrew Fraser, a professor at the University of Toronto's Donnelly Centre for Cellular and Biomolecular Research, said while anastasis—which he hadn't heard of before—could mean good things for ischemia or strokes, it could also be dangerous.
Advertisement
"Typically when you trigger a cell to die, you really want it to die. It's had massive DNA damage or something," he said. "From the perspective of the organism, if I wanted that thing to be dead, I really need it to be dead."To him, the most important issue is the extent of DNA damage. What happens if the cell starts to die, but goes on to develop mutations through anastasis?"Obviously what you don't want to do is to pass that on," he said. "It's very well known that apoptosis is a major outcome of DNA damage—that's the organism basically going, 'Oh my god my DNA's been hit,'" then eliminating cells to limit damage.According to Montell, Fraser is right to worry: in her observations, a small fraction of cells retained DNA damage and mutations, which could lead to cancer. But most cells undergoing anastasis can repair their DNA damage, she said. "An unfortunate side effect of retaining that capability, to bounce back, is that in a minority of cases they're going to mutate a gene."The second time I spoke to Montell, she was away from her California lab at a conference in Texas. Her team had just released more information in a paper, published to a preprint server for now, which shares new observations: that anastasis happens in two stages; that cells undergoing anastasis multiply, then migrate; that after 12 hours, cells aren't quite back to their original condition (leading researchers to wonder if cells would ever be normal or if anastasis leaves them permanently changed). These processes are similar to a wound-healing response, Montell said.
Advertisement
But doesn't that also sound eerily similar to cancer—to multiply, to migrate (metastasize), to come back despite exposure to toxins?Well, yes. This is exactly what you don't want cancer cells to do, Montell said. "When we treat cancer, we treat it with radiation or chemotherapy—and what that does is cause cancer cells to commit suicide by apoptosis."Cells that bounce back from the brink of death could explain relapses. Months or years after successful cancer treatment, cancer can come roaring back. According to Montell, anastasis could be responsible. "The cancer cells are hijacking the normal, positive repair process and unfortunately when cancer cells do it, it has a negative outcome," she explained. Cancer cells are just doing what normal cells do—trying to heal and migrate.If it turns out that anastasis is the process responsible for cancer coming back after remission, she said, it would be "an unfortunate byproduct" of a process the researchers assume evolved to heal tissues after severe but sublethal injury. "That's not a brand new idea: cancer as a wound that won't heal, or an ongoing, never-terminating wound-healing response."Get six of our favorite Motherboard stories every day by signing up for our newsletter.
